Ground-breaking finding for Colorectal Cancer! Disulfiram’s New Role in Precision Oncology
An old drug for alcoholism could transform the way colorectal cancer is treated - from underdog repurposed agent to precision metabolic killer
If you have colorectal cancer, this is essential reading! Laboratory research has uncovered an exciting vulnerability that could transform treatment outcomes.
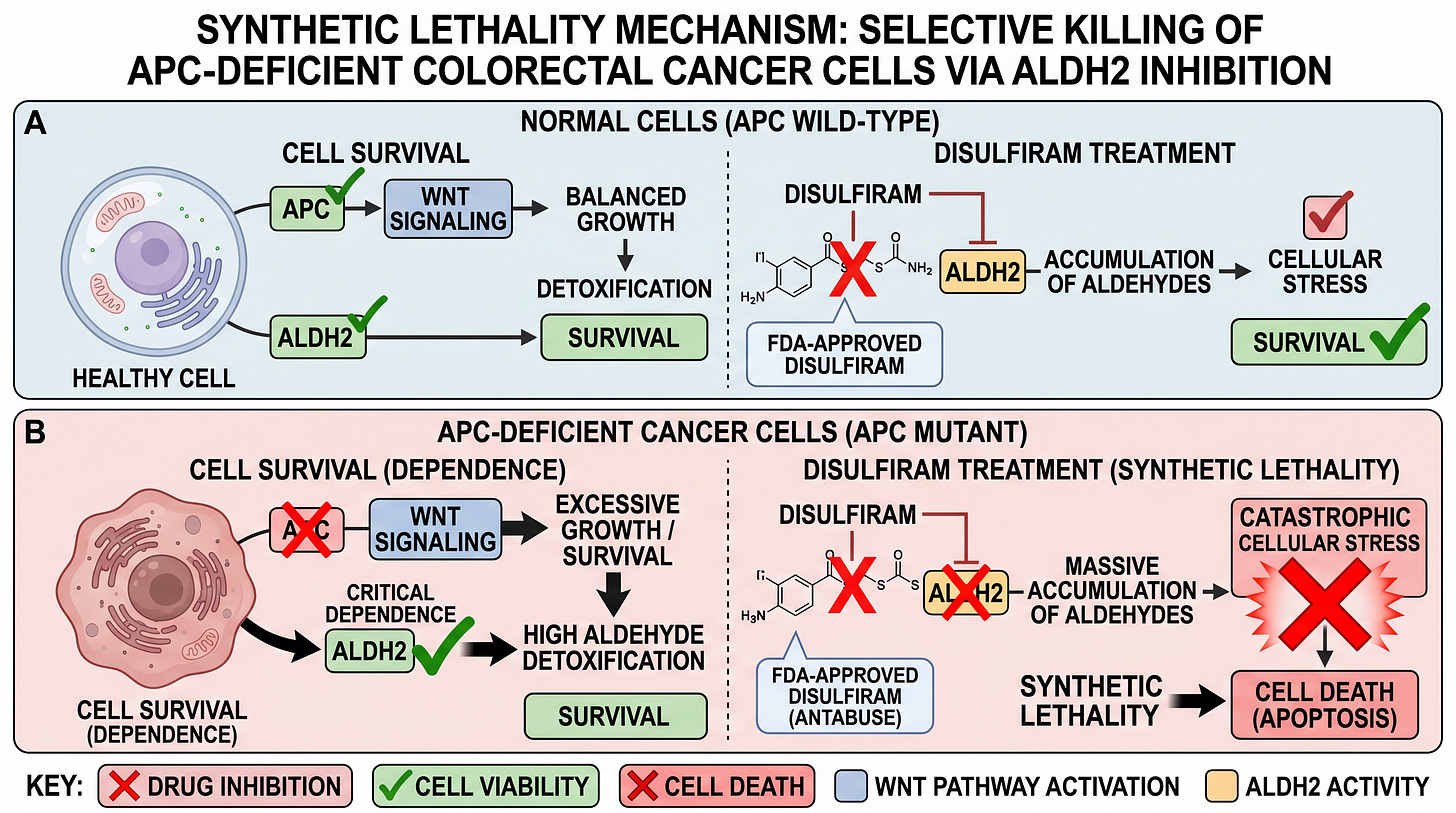
The discovery centres on a common mutation in the APC gene. These mutated cancer cells become unusually dependent on an enzyme called ALDH2 to survive. ALDH2 has been explored for a long time in the field of cancer. Researchers have been eyeing disulfiram (as I have for over 10 years) as a potential treatment for many cancers. I think it could help create a condition similar to ferroptosis called cuproptosis — where copper rather than iron is the key to a Kill Phase “pulse.”
This old, inexpensive drug called disulfiram, originally used to treat alcohol addiction, strongly blocks this enzyme (ALDH2), and that can push the cancer cells into fatal oxidative stress — overwhelming their antioxidant protection. This genetic-specific effect could turn disulfiram from a “maybe it helps” repurposed drug into a TARGETED THERAPY for the majority of colorectal cancer patients who carry APC mutations. Amazing! Honestly, this is BIG, but I have not seen it mentioned in any of the national papers; even follow-up reports in the medical press don’t mention disulfiram either — almost as if they are hiding it.
The APC Mutation: A Major Driver in Colorectal Cancer
Colorectal cancer (CRC) is one of the most common cancers in the world, with nearly 2 million new cases diagnosed each year and a horrifying increase in rates among younger patients.
At the heart of many of these tumours is a problem with the APC gene, a natural “brake” on cell growth located on chromosome 5, which is linked to tumorigenesis. When APC is mutated or lost (which happens in roughly 80–90% of non-inherited colorectal cancers), it causes the Wnt signalling pathway to go into overdrive. There are several repurposed drugs that target this “undruggable” Wnt pathway — but let’s leave that for another time.
The Wnt pathway then leads to uncontrolled cell division and tumour reactive oxygen species (ROS) as a by-product. To cope, the cancer cells lean heavily on antioxidant systems such as glutathione and the enzyme ALDH2, which helps clean up toxic by-products from damaged fats (lipid peroxidation).
New preclinical research published in March 2026 shows that APC-deficient colorectal cancer cells are already “on the edge” because of this extra oxidative stress. They depend far more on ALDH2 for survival than normal cells do.
Synthetic Lethality: A Smart Way to Kill Cancer Selectively
This situation creates what scientists call synthetic lethality — a weakness where blocking one thing (ALDH2) is harmless to normal cells but deadly to cells that already have the APC mutation. It might be worth a post on this topic alone, as synthetic lethality — this two-punch hit to the cancer cell — is really important.
Disulfiram fits the role perfectly. It’s an FDA-approved drug (sold as Antabuse) that has been used since the 1940s. If you take it and then drink, you will feel really nauseous with a banging headache. This is because it blocks aldehyde dehydrogenase enzymes (ALDH2).
Disulfiram’s Long Road from Alcohol Treatment to Cancer Research
For decades, researchers have explored disulfiram’s potential against cancer, especially because many cancer stem cells rely on ALDH enzymes. In colorectal cancer studies before 2020, disulfiram (often combined with copper) showed it could kill cancer cells by disrupting the proteasome, causing stress in the cell’s protein factory, and triggering immune responses.
Earlier work was fairly broad — trying to kill cancer cells without focusing on specific mutations. But the 2026 study changes all of that: it shows disulfiram works particularly well because it exploits this unique weakness created by APC mutations.
How It Actually Works: Overwhelming the Cell with Stress
In lab models using APC-mutant colorectal cancer organoids (mini-tumours grown in a dish) and mouse xenografts, disulfiram dramatically reduced cancer cell survival — often by 50–70% on its own, and nearly completely when combined with a bit of copper.
Here’s why: APC-mutant cells already produce extra toxic aldehydes from fat damage in carcinogenesis. ALDH2 normally mops these up. When disulfiram blocks ALDH2, the toxic aldehydes build up, ROS levels (free radicals) spike, and this activates a stress pathway (ASK1 → JNK) that tells the cell to die through apoptosis (programmed cell death).
In mice with APC-mutant tumours, the disulfiram + copper combination shrank tumours by about 60% without causing obvious harm to the animals’ normal tissues. Importantly, cells without the APC mutation were hardly affected — highlighting the selective nature of this approach.
Why This Is So Important
APC mutations are extremely common in colorectal cancer:
They occur in 60–70% of tumours on the right side of the colon.
They’re present in nearly all cases of familial adenomatous polyposis (FAP).
These mutations can be easily identified with standard genetic testing (next-generation sequencing) or even certain pathology stains.
Disulfiram has big practical advantages:
It’s generic and very cheap (around £15 per month).
It’s taken as a pill.
It has a long safety record in humans.
Side effects are generally mild (mainly nausea or mild nerve issues), especially if patients avoid alcohol.
It could be combined with standard chemotherapy (like 5-FU), immunotherapy, intravenous vitamin C, or other metabolic drugs to make treatment even more effective.
How It Stacks Up Against Current Treatments
Current metastatic colorectal cancer treatments are often expensive, toxic, and not selective for specific mutations. Disulfiram stands out for its low cost, mild side effects, and potential genetic selectivity:
FOLFOX: DNA-damaging chemo — ~40–50% response rate, significant neuropathy and low blood counts, costs thousands per month
Targeted drugs (e.g., for BRAF mutations): effective only in small subsets, very expensive
Disulfiram + copper (preclinical data): high activity in APC-mutant models, mild side effects, costs under £50/month
Looking Ahead: Time for Clinical Trials
This discovery opens the door for smarter, mutation-guided use of an old drug. The next logical steps are clinical trials specifically in patients with APC-mutant colorectal cancer — testing disulfiram (with or without copper) added to standard treatments.
Patients and advocates can help by:
Asking doctors about APC mutation testing
Supporting repurposed-drug research (remember to subscribe and SHARE!)
Pushing for basket trials that match treatments to genetic profiles
In lower-resource settings, an affordable, oral option like disulfiram could make a meaningful difference where expensive biologics are out of reach.
Clinical Relevance: Why APC-Deficient CRC Screams for This Approach
APC mutations define CRC subsets: 60–70% in proximal tumours, 80% in FAP-associated cases. They’re detectable via standard NGS panels (e.g., MSK-IMPACT) or IHC (nuclear β-catenin). Patient selection is plug-and-play — no new biomarkers needed.
DSF’s profile shines: oral, £17/$20 per month generic, half-life ~10 hours, metabolised to diethyldithiocarbamate (DDC), which chelates copper.
Phase I oncology trials (e.g., NCT03323346 in glioma) confirm safety up to 640 mg/day, with copper supplementation feasible.
Toxicity? Mild (nausea, neuropathy); avoid in alcohol users or Wilson’s disease.
Combo potential abounds: pair with 5-FU/leucovorin (standard adjuvant) to hit proliferating APC-mutants. Or immunotherapy — DSF’s ICD boosts PD-1 efficacy. Metabolic synergies? Metformin (AMPK activator) or statins (mevalonate blockade) amplify ROS.
Barriers? Bioavailability — DSF’s short half-life demands thrice-daily dosing; liposomal or Cu-ET formulations (in trials) could solve this. Also, APC–wild-type resistance: only 20% of CRC, but stratify to enrich responders.
Wrapping Up the Promise
This ALDH2–APC synthetic lethality isn’t hype — it’s mechanistically sound, genetically precise, and clinically actionable. Disulfiram evolves from underdog repurposed agent to precision metabolic killer, potentially benefiting most CRC patients. With trials underway for DSF in oncology, 2026 could mark the pivot. Stay tuned; science delivers when metabolism meets genetics.
This isn’t just another “drug might work” story. If you have an APC mutation, it’s huge. It’s a clear example of precision metabolic targeting — looking at the cancer’s biology as well as the mutations, finding the specific weaknesses created by a common cancer mutation to design smarter, gentler treatments.
If upcoming trials confirm these findings, disulfiram could evolve from an old alcohol-aversion pill into a genuine targeted therapy for the majority of colorectal cancer patients. Science will make a huge leap forward when genetics and metabolism meet. This could be one of those moments.
Let’s see if it makes the national papers.
I

It’s encouraging to see repurposed drugs make their way into cancer research. I’ve written myself about how I used several repurposed drugs in my own journey and remission. An important thing patients often miss is that precision matters. The same drug can behave very differently depending on the cancer type, mutations, metabolic profile and treatment phase. That’s why NGS and other types of genomic testing are so necessary these days to become part of routine testing in cancer.